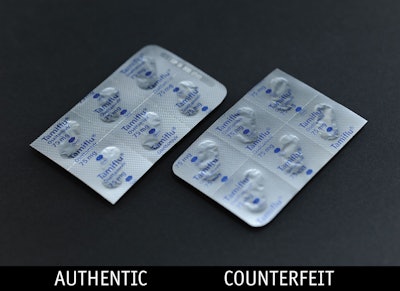
As the U.S. withdraws combat forces from Iraq and Afghanistan, the battle rages on against counterfeit products and packaging around the globe.
Havocscope, an online database of black-market activities, says, “In today’s interconnected world, black markets have the power to wreck havoc on all industries and across all regions. By denying needed tax revenues from governments and destabilizing society and the environment, the global black market impacts everyone on a daily basis.”
Havocscope pegs the financial value of global counterfeit markets at $1.92 trillion, with the average country counterfeit market at $21.16 billion. The U.S. tops the database’s list of 91 countries in terms of its counterfeit market value at $621 billion, followed by China, Mexico, Japan, Spain, and Italy.
It estimates the financial value of global trade in counterfeit goods at $585.38 billion. In ranking 28 products/goods categories, the organization ranks counterfeit drugs first at $200 billion, followed by counterfeit electronics at $100 billion, and then software piracy, foods, auto parts, toys, movie and movie piracy, clothing, and shoes. Sporting goods, tobacco, cosmetics, alcohol, pesticides, and money are also on the list. Havocscope’s Web site also presents the following details:
-
Of 11 million mobile phones in the United Arab Emirates, 70,000 are counterfeit.
-
In 2010, the Anti-Counterfeiting Group of the U.S. Golf Manufacturers reported that 25,000 pieces of counterfeit golf equipment, such as golf clubs, bags, shoes, and balls, were seized in raids conducted across major cities in China.
-
A U.S. Senate investigation in 2011 found about 1,800 incidents of counterfeit electronic parts being sold to the U.S. military. The incidents involved over 1 million counterfeit parts, with 70% of the counterfeit items traced back to China.
Counterfeit product expansion
Jim Rittenburg, chair of the International Authentication Assn, (IAA), put the global counterfeiting scenario into perspective in a June 30, 2010 online question-and-answer interview in which he explained: “It used to be taken for granted that it was only luxury and high-end goods such as perfumes, spirits, and designer clothes that were most at risk from counterfeiters and pirates. However, that’s no longer the case with counterfeiters—aided by advances in technology—turning their attention to any branded item that will generate a profit. As a result, everyday household items including foodstuffs, cigarettes, beverages, and hardware items are equally targeted.
“One of the effects of globalization is that emerging areas such as India, China, Russia, and parts of Africa have become black spots for the production of sub-standard goods. The danger is that without adequate policing and checks, these goods can enter the supply chain and impact the safety and security of existing brands.”
The IAA online article noted that the Organization of Economic Development (OECD) estimated the global counterfeiting market topped $200 billion, while the Counterfeiting and Intelligence Bureau (CIB) predicted fake goods make up to 7% of world trade.
IAA estimated that U.S. businesses lose $200 to $250 billion a year due to counterfeiting, with 750,000 jobs lost in this country and 100,000 in the European Union. The timeframe on these statistics is unclear.
IAA also reported that 10% of drugs sold in the U.S. are fakes, citing the U.S. Food and Drug Administration. Referring to 2007 Army AL&T data, 36,000 fatalities and 1.5 million accidents on U.S. roads were attributed to defective automotive parts.
‘Crimes of opportunity’
“Let’s be honest,” says Wes Shepherd, CEO of online monitoring firm Channel IQ, “industry will constantly be combating counterfeiting.” He adds, “Packaging has a very important role, but ultimately industry will have to keep coming up with other technologies. The biggest problem I see is that packaging is no longer the key to ensuring product legitimacy for gray market and counterfeit concerns. With the Internet, people can represent a product as legitimate and there are few ways to guarantee its legitimacy.”
The situation is not acceptable to Margaret Hamburg, M.D., the FDA’s commissioner of Food and Drugs. In remarks at the Partnership for Safe Medicines Interchange 2010, she described counterfeiting and diversion as “crimes of opportunity.” She noted, “Drug counterfeiting, diversion, cargo theft, and economically motivated adulteration are crimes of opportunity, and the opportunity is flourishing because of the dramatic way our world has changed in a relatively short period of time. In the past decade or so, the pharmaceutical industry has shifted a large part of its manufacturing operations and supply sourcing overseas. Today, nearly 40 percent of the drugs Americans take are imported and nearly 80 percent of the active ingredients in the drugs on the American market come from overseas sources.
“So, in addition to the growth in volume of imports, there has been a dramatic increase in the variety and complexity of imported products. As a result, the supply chain—from raw material to finished product—has become more complex and mysterious involving a web of repackagers and distributors in a variety of locations. Like any chain, the drug supply chain is only as strong as its weakest link, and the proliferation of additional handlers, suppliers, and middlemen creates new entry points through which contaminated, adulterated, and counterfeit products can infiltrate the drug supply. And this, in my view, is simply unacceptable.”
USP guidance to come
During September’s 9th Cold Chain & Temperature Management Global Forum in Philadelphia, Dr. Mary Foster, chair, packaging and storage expert committee with the U.S. Pharmacopeia (www.usp.org) and vp with Aphena Pharma Solutions, noted that new guidance in USP’s General Chapter <1083> has “the ultimate goal to prevent adulterated and misbranded materials from entering the supply chain and to detect counterfeiting or diversion.”
Says USP, “As the pharmaceutical supply chain grows increasingly complex, and the risks to drug quality and safety correspondingly proliferate, new draft guidelines will be proposed [in] spring by the U.S. Pharmacopeial Convention to help ensure the integrity of the supply chain. A significant component of the guidelines, which will be contained in General Chapter <1083> of the USP-NF compendia, is best practices to combat counterfeit drug and medical devices.
“The chapter notes that combating counterfeit drugs and medical devices requires a combination of strategies that involve the cooperation of pharmaceutical or device manufacturers, supply-chain entities, regulatory authorities, law enforcement, and patients. No single approach or party can be successful working in isolation. Within the chapter, USP looks at topics including packaging technologies (tamper-evident designs, authentication technologies, and serialization); establishment of drug pedigrees; application of machine-readable data carriers to drug products (2D bar codes and RFID tags); repackaging guidance, information retention, and security; international standards and global approaches to establishing drug pedigrees; combating illegal Internet pharmacies; and best anti-counterfeiting practices.”
The chapter also covers good importation practices, diversion and theft, and natural disasters. The information will be published in the Pharmacopeial Forum, the vehicle through which USP accepts public comment on its standards, in the March-April 2012 timeframe. This is available free to all parties. USP expects the chapter to evolve significantly based on public feedback, and will further solicit input at a Supply Chain Integrity Workshop that the organization is convening May 22-23, 2012, at its headquarters in Rockville, MD.
Pharmaceutical focus
IAA reported, “All sectors are affected in one way or another. In many of these cases, protecting the integrity of brands can be the difference between life and death.” That’s particularly the case with fake pharmaceuticals, where financial opportunities increase the possibility of counterfeiting and diversion.
The International Policy Network US Inc. (www.policynetwork.net), a nonprofit organization that seeks to bring down barriers to enterprise and trade, reported in 2009, “approximately 700,000 people die annually from fake malaria and tuberculosis drugs alone. The report lays bare the ballooning problem of counterfeit and substandard drugs, which can constitute one third of the drug supply in certain African countries. These dodgy drugs result in unnecessary death and increased levels of drug resistance."
New technologies make it more possible to identify counterfeiting and diversion around the globe. Take Africa, for instance. In mid-October, telecommunication provider Orange Kenya launched mPedigree, an SMS-based system to fight counterfeit drugs in Kenya and Cameroon, where up to 30% of the drugs are reported as potentially affected.
Plans call for this system to be rolled out in different healthcare centers all over the country. Every drug serial number will have to be scanned through to ensure it is legitimate before the drug is administered to any patient. The announcement to launch mPedigree into the operations of Orange across the continent was made earlier last year in Nairobi, following a partnership between the telecommunication services provider and the initiators of mPedigree. Orange Kenya is already working with Qualcomm, a driver behind next-generation mobile networks, devices, applications and services, as well as with other partners in a project called “Wireless Reach,” which is involved in the automation of the procurement of Anti-Retroviral Drugs at public clinics.
Also last year, Systech Intl. (www.systech-tips.com) announced that with its Brazilian partner Videojet (www.videojet.com), one of the world’s largest pharmaceutical manufacturers would implement Systech’s Serialized Product Tracking (SPT) on each of its packaging lines in Brazil to meet that country’s pending anti-counterfeiting track-and-trace requirement, Law 11.903. The law, passed in 2009, requires the “tracking of manufacturing and consumption of medicine by means of data capturing, storing, and electronic transmission technology.”
SPT allocates and verifies unique identification numbers at the item level, packaging the item (child) into a higher-level package (the parent), establishing a relevant parent-child relationship between item, case, and pallet, and maintaining data integrity throughout the packaging process, ensuring the manufacturer’s serialization and aggregation data are accurate.
In August, multilayered brand protection supplier Covectra and Belfast, Ireland-based carton and label provider MSO Group announced a partnership to provide serialization for track-and-trace and diversion control, offering unit-level product identification through variable-data printing of 2D bar codes and human-readable codes.
These technologies aim to address Europe’s Falsified Medicines Directive, which requires authenticity features on prescription drug packaging. “This newly developed directive by the European government is a first step in protecting consumers and saving pharmaceutical companies millions,” says Steve Wood, Covectra’s president. “The initiative will certainly put pressure on the U.S. government to continue to aggressively address what is now being referred to as an epidemic.”
4 Serialization team
Made up of suppliers Acsis, Cognex, Nosco, and Omega Design, the 4 Serialization team was developed to provide a source for serialization and e-Pedigree solutions for pharmaceutical and life sciences organizations to provide a safe and secure supply chain to comply with government mandates and protect patients.
The team helps companies achieve compliance with the approaching e-Pedigree and serialization mandates, building a solution flexible enough to meet individual needs and to support changing mandates. It works with serialization suppliers to meet serialization requirements while collecting the data necessary to build and benchmark ROI, engineering packaging with preserialized 2D and RFID tags, and launching structured pilot programs and production scale-ups, maximizing results without affecting machine speeds and accuracy.
Pedigree to the patient
Clearly, packaging technologies continue to become more sophisticated to take on counterfeits. Overt and covert technologies, e-Pedigree serialization, and global plans continue to be developed. Yet these technologies rarely seem to reach to the patient.
One of the leading advocates of involving the patient in the supply chain is Walter Berghahn, president of SmartRmeds for Life (www.smartrmeds4life.com) and executive director of the Healthcare Compliance Packaging Council.
Berghahn believes, “The U.S. is the only country in the world still clinging to the antiquated practice of repackaging drugs in the pharmacy. This practice had a place 100 years ago in the world of the compounding pharmacist. It has no place in our modern world of global manufacturing in state-of-the-art facilities with packaging that has been designed and tested to protect the specific product being carried to market. If we truly want to end-to-end supply-chain security, then the patient should receive the original manufacturer’s container and have the ability, via smartphone or the Internet, to authenticate that ‘Yes, this container was manufactured by pharma company X and this is a real container.’”
On its Web site, the U.S. Department of Commerce offers its “Top 10 Ways to Protect Yourself From Counterfeiting and Piracy.” The top step listed: Scrutinize labels, packaging, and contents. “There is no foolproof way to know the difference between a bargain and a fake, but labels and packaging can be revealing indicators,” says the site. “Look for missing or expired ‘use-by’ dates, broken or missing safety seals, missing warranty information, or otherwise unusual packaging. …You can also verify authenticity by comparing the manufacturer’s contact information with another product’s packaging, as addresses and phone numbers provided with counterfeit goods could be misleading.”