From what we've been seeing in technology developments, there's a trend toward vendors providing containers and materials already embedded with RFID inlays. It's a trend that packagers hope will bring costs down for them. That was one of the desires expressed during a recent Packaging World panel discussion on radio-frequency identification (RFID) that will be presented in our upcoming December Special Report on RFID. Participants included RFID experts from ConAgra and Tanimura & Antle.
Lower pricing results from the use of smaller RFID inlays versus the larger RFID tags commonly used on containers and usually applied in production by the packager.
Another packaging container supplier expanding into this area is O-I Healthcare Packaging Group, Perrysburg, OH. We recently spoke with Brian Chisholm, project engineer for the RFID program, and Ryan Gladieux, business development manager-pharmaceuticals.
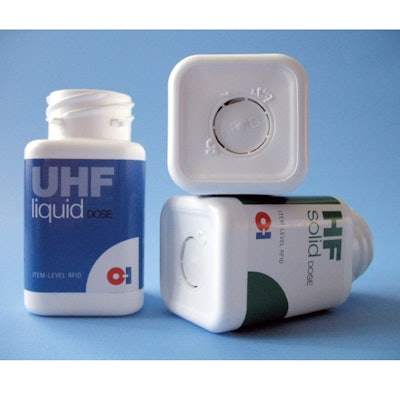
O-I is starting with white pigmented 75-cc square bottles molded of high-density polyethylene, it's most popular size, though Chisholm notes they can apply the technology to polypropylene bottles, too. The opaque bottles are commonly seen on shelves behind pharmacists' counters.
O-I's research efforts into RFID embedding began one year ago. According to Chisholm, O-I is working with companies such as Tagsys, Impinj, and RSI ID Technologies.
Small as a nickel
The inlays O-I uses are 17-mm dia, just larger than a nickel.
Chisholm points out the fact that embedding makes the tags tamper-evident. "The only way to remove a tag is by somehow destroying the bottle," he says. "Someone must cut into the container to get the tag out."
O-I embeds the RFID tags centered in the base of the bottle, parallel to the bottom.
"This kind of consistent orientation relative to the bottle improves readability on a line," Chisholm claims. "RFID-enabled labels may contact each other, and sometimes that interferes with the tags' readability. Also, post-labeling damage of the RFID tag can occur if bottles hit each other. Recall that a silicon chip is basically made of glass. We believe that once embedded in the package, tags are much more robust than applied as part of a label."
HF or UHF
O-I can use either HF or UHF technology and maintains RFID inlays for both frequencies. It is also making use of Impinj's near-field UHF design, which Chisholm feels deserves serious consideration for item-level applications.
Chisholm says O-I is nearing commercialization, and will move into pilot scale production by the end of 2006. That will permit it to pinpoint product pricing, Chisholm says. Until then, he says that "Embedded containers will be very competitive with system costs for RFID labels."
O-I added ancillary equipment to assist with embedding, "though the changes are nothing dramatic," says Ryan. "It's a process similar to in-mold-labeling and doesn't slow down our speeds at all."
O-I is pleased that it can use an RFID inlay rather than a converted RFID label, which is typically larger and more costly.
Better than embedded closure?
Chisholm feels that a tag embedded in the bottle is better than in the closure, a method that has been made available by companies including Tagsys and West Pharmaceuticals (see RFID-enabled vial seal, published June 2005 or a more recent mention, in our November 2006 issue, California dreamin'.)
"If the tag is in the bottle, then it cannot be removed or swapped inadvertently or otherwise," Chisholm points out. 'It's a better solution from a package integrity standpoint to have tag and bottle integrated." An RFID tag in the closure liner is susceptible to tampering, he adds, though electronic tampering prevention is part of the security of the Electronic Product Code portion of the tag memory.
"A person cannot modify the tag's identification number. It's like an engraved license plate," says Chisholm. "It's a pretty secure system."
In the next development stage, O-I is working on a project to test and compare HF and UHF "in our customers' packaged dosage forms," Chisholm says. "This should help our customers understand the differences between the two and dispel or validate claims especially that UHF does not read around liquids or metals."
We reported on a White Paper that addresses the technical issues of UHF vs HF RFID, UHF benefits updated, which appeared in our October 2006 issue.
O-I, which maintains that choice is up to its customers, is currently seeking a customer to partner with for a pilot.
Successful demo announced
The company is demonstrating its RFID technology publicly for the first time November 13-14 at the RFID HealthCare Summit in Washington, D.C.
Update: In the demonstration, a near-field UHF Gen 2 RFID solution, comprising Impinj's Speedway™ reader and O-I's item-level embedded RFID tags powered by Impinj's Monza™ chips, achieved 100% reliability when reading and writing tags at high speed on pharmaceutical bottles.
In the first demonstration, the UHF Gen 2 system programmed 96-bit EPC numbers at rates exceeding 600 tags per minute on bottles containing the four most common dose types — liquids, gel caps, solids and powder. In the second demonstration, the system flawlessly read 600 tags per minute on cases containing 48 individually tagged bottles and a case-level tag.


























