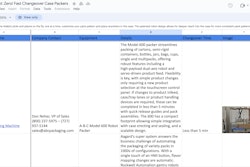
Starting in 2010, "all pharmaceutical cartons (in the UK) will be required to include Braille information." Braille-embossed cartons and labels on secondary drug packaging have been required in the EU since 2006. To that end, several packagers are devising a variety of ways to provide compliance: tooling of cartons, special adhesive and varnishes, and shrink-sleeve technology. Visit www.packworld.com and www.healthcare-packaging.com for information on specific solutions.
Source: www.printweek.com/
Source: www.printweek.com/