Medication nonadherence/noncompliance, where patients fail to take their medications as prescribed, is contributing to “a global epidemic causing countless deaths and billions of dollars in health care system waste annually,” according to The Medication Non-Adherence Blog.
And how’s this for alarming: “Although around 30 percent of nonadherence can be attributed to unintentional behavior, such as forgetting, a staggering 70 percent of nonadherence is actually intentional, according to Professor John Weinman, HOD of Health Psychology at Kings College, London.”
That’s according to Kate Reid of Atlantis Healthcare, whose article, “The Heart of the Non-Adherence Epidemic,” appeared in The Medication Non-Adherence Blog earlier this year. Reid’s blog says, “Tackling nonadherence to medicines could be the single-greatest innovation in reducing wastage and improving health outcomes for all. Nonadherence is a complex behavior, driven by the beliefs that patients have about their condition and their treatment.”
Asked to comment on the 70% quote regarding intentional nonadherence, Walter Berghahn, president of SmartRmeds for life, and executive director of the Healthcare Compliance Packaging Council, offered the following insights:
“It's a fair statement, but I think the level may be high, although, if you include folks who cannot afford their meds and ‘choose’ not to buy them, then maybe it could be valid that 70 percent of nonadherence is intentional. That being said, poor health (medication) decisions often come from a lack of knowledge.
“The HCPC always promotes that graphics on compliance packaging are not solely about reminding but equally important, educating. Lack of understanding for the disease condition or asymptomatic conditions from high cholesterol or high blood pressure are a huge problem. Packaging alone will not solve this. Reminders and educational tools in print, electronic form, and social media all have a role in improving adherence.
“The packaging has the unique benefit of staying with the product and has the best chance of being seen by the patient and family members on a daily basis. This is why we feel it is so important to start with the package and build outward with other tools that link to or key off the package. Calendarized packaging has the benefit of leaving evidence of the dispense events in the simplest form—Tuesday’s [dose] is there or it is not. Short of integrated electronics to detect opening events, a simple calendar feature is right in the face of patients and caregivers (family or professional) on a daily basis.”
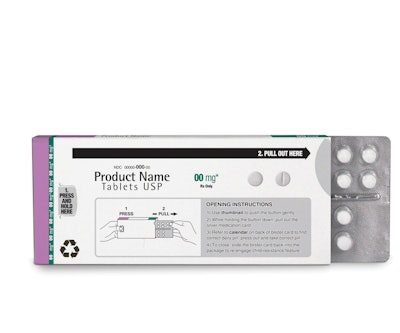
Walmart to launch new compliance pack
Big news on the compliance-prompting packaging front came when Walmart announced that this summer, more than 3,600 of its nationwide pharmacies would employ the new Shellpak® Renew package from MeadWestvaco Corp. Shellpak Renew features a tear-resistant, recyclable outer carton, an easy-slide blister, and an integrated calendar for patients to easily track their medications. (A sample retail pack is shown here.)
“Medications prepackaged in Shellpak Renew make dispensing medications quicker and easier so that our pharmacists can spend more time counseling our patients, including providing instructions on how to take their medications,” notes Sandy Kinsey, vice president, Pharmacy Merchandising at Walmart Stores. “In addition, the calendarized packaging helps reinforce to our customers how to take their medications safely and effectively for the best outcome.”
Kinsey adds, "Our packaging initiatives are principally focused on the patient and the ability to improve therapy adherence by properly taking medication as prescribed. It is our hope that the industry will join us in these efforts and eventually migrate away from traditional vials for the benefit of the customer."
MWV cites research showing that half of all patients in the U.S. today do not take their medicines as prescribed. Poor medication adherence can lead to worsening of disease, serious and avoidable health risks, increased hospitalizations, and death. The annual cost of nonadherence in the U.S. is estimated to be $290 billion, representing 13% of total healthcare expenditures.
Shellpak Renew is based on MWV’s original Shellpak design, and was developed with feedback from pharmacists and patients. The new Renew packaging has a smaller footprint requiring less shelf space. The outer carton is made of MWV’s Natralock®, designed as an environmentally friendly paperboard-based packaging that maximizes the use of recyclable materials and minimizes waste. The package also contains enhanced adherence features, including color-coded spine labels to help distinguish among drugs, and easy-open, child-resistant push buttons.
.
Part of the healthcare ‘solution’

























