After receiving U.S. Food and Drug Administration approval in October 2008, Pfizer launched its overactive bladder (OAB) treatment Toviaz™ in a carded blister housed in a paperboard outer sleeve and bundled with regulatory literature for presentation to consumers by prescribing physicians.
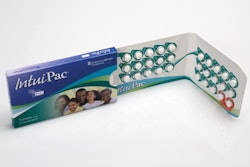
Last year, Pfizer worked with AmerisourceBergen company Anderson Packaging to transition to Anderson’s proprietary IntuiDose™ child-resistant compliance-prompting format for the physician’s sample package. The four-panel wallet pack contains 14 8-mg doses. The user pushes each pill out of each blister cavity from a three-ply cold-formed base film. The lidding is a 15#, paper-supported, push-through 0.008 foil. The pack uses a 16-pt board stock, coated on two sides (C2S), with a heat-seal coating applied on press during printing to help reduce overall cost and material use. Also, there are 4-mg and 8-mg strength versions, distinguished by an orange or green color scheme.
The Toviaz pack recently was honored as the 2009 Compliance Package of the Year by the Healthcare Compliance Packaging Council, and as the first Distribution Management Award-winner by the Healthcare Distribution Management Assn.
The new pack allows Pfizer to deliver Toviaz in a more effective and informative patient-friendly format, at the same time achieving substantial production efficiencies and cost reductions.
The package includes a daily tracker, functioning as a guide and diary to track goals and health improvement. The program also provides a tool to review progress with the physician over the 12-week course of treatment. A customer response card is integrated in the pack, allowing patients to complete a questionnaire and enroll in an interactive treatment program to ensure success. Finally, the pack includes full patient prescribing information. Tablets are laid out in a simplified 14-day regimen, including instructions about how to properly ingest the medication.
The IntuiDose package features a novel approach to providing the highest levels of child resistance while at the same time making the product easily accessible to adults. The simplified push-through design focuses on providing a consumer-friendly solution to medication management. The use of a unit-dose compliance-prompting package facilitates medication adherence and effectiveness of treatment, guiding patients to fill their prescription and maintain compliance over the course of treatment.
Production/packaging efficiency gains
According to Anderson Packaging, the transition to the IntuiDose design provided opportunities for gains across the Toviaz supply chain for Pfizer, most notably in packaging operations. The production of the initial Toviaz sample design involved a multiple-stage packaging system. Packaging was initiated with the primary packaging of the Toviaz tablets into a cold-formed blister design. In a second operation, blisters were heat-sealed into paperboard cards. Cards were then loaded into paperboard sleeves, ensuring the activation of the child-resistant feature. In a final operation, completed packages were bundled with patient literature and loaded into shipping containers for distribution.
Production of the improved Toviaz design utilizing the IntuiDose delivery system substantially reduced product handling through the simplification of the package assembly and incorporating highly automated packaging systems.
Like the original assembly, the process begins with blistering of the tablets. The improved IntuiDose design assembly reduces overall packaging operations to two steps. Upon the completion of the blistering operation, blisters are heat-sealed into a wallet format. Each piece of marketing and regulatory literature is applied to the package in an integrated process, with high-speed gluing and verification. The improvement in package design reduces material handling, decreases material consumption, minimizes the generation of scrap, and ultimately reduces processing time, resulting in logistical efficiencies.