We knew from a sales and marketing standpoint that if the technical challenges could be met, this package would be a success.”
That’s how director of marketing Shari Sandberg describes the mood at B. Braun Medical Inc. when the Irvine, CA, firm came up with the Duplex® drug delivery system, a new package for intravenous drugs whose liquid and powder components must be kept separate until they’re administered to the patient.
Partnering with B. Braun in building the unique aseptic packaging system behind the Duplex package was Italy’s IMA, represented in the United States by IMA North America (Bristol, PA).
IMA adapted its reliable powder- and liquid-filling technology to B. Braun’s unique needs and then put the whole system together inside isolator cabinets. “We could have taken a more conventional approach, where machines are in a clean room and operators are gowned up,” says Sandberg. “But that involves too much human intervention. With the isolator approach, we shrink the room around the machines and keep operators out. The only access to the equipment is through glove ports.”
Each isolator is a stainless steel and glass enclosure. Above it is an air-handling system with HEPA filtration that ensures a positive pressure is always maintained inside, thus providing a contamination-free environment.
Not only is there one isolator cabinet for the powder-filling machinery and a second for the machine that fills the liquid diluent. A third is used to decontaminate the trays in which premade Duplex containers arrive at the Irvine plant, and a fourth is used to fill the hopper of the powder filler from the bulk drug container. These last two isolators make it possible to open the trays of Duplex containers and the bulk container of powder inside a sterile environment. Bioquell Pharma (Andover, Hampshire, England) built the isolators.
Addressing a need
As with so many packaging breakthroughs, B. Braun’s development of the Duplex concept began with an analysis of where current technology left room for improvement. That technology consisted typically of a glass vial for the antibiotic powder and a bag for a diluent. Health professionals would add the powder to the diluent, mix them together, and then attach an IV drip to the bag. This represented too much labor and too great an opportunity for an error in mixing.
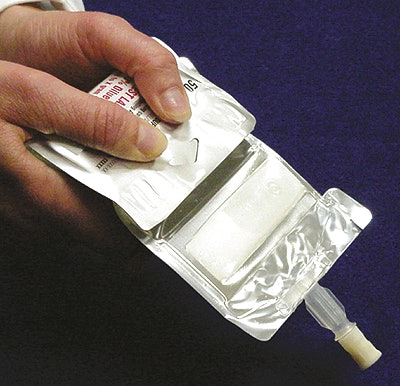
The solution B. Braun came up with is a 4”x7” flexible bag consisting of three compartments each separated by a peelable seal. At one end is a delivery port attached to a compartment that’s empty. Above it is a small compartment that contains 0.5 to 2.0 g (.018 to .071 oz) of antibiotic powder. And above that is a large compartment holding as much as 100 mL (3.38 oz) of diluent.
To administer the drug, the nurse squeezes the upper compartment holding the diluent. This bursts the first peelable seal and mixes the diluent with the powder in the middle chamber. Once the two are sufficiently mixed, the nurse squeezes the second chamber until the second peelable seal breaks. That puts the product in the chamber with the dosing port. All that remains is connecting the port to the patient’s administration tubing and hanging the bag from an IV pole for delivery.
B. Braun makes the package in its own established IV bag-making facility on its Irvine campus. The firm acknowledges that its abundant in-house bag-making experience gave B. Braun a valuable head start in designing the Duplex drug delivery system.
The Duplex container is fabricated from several materials. The idea is to protect the powdered drug from oxygen and moisture and to protect the diluent from water vapor transmission.
The labeled front side of the container is a foil laminate consisting of PET, foil, and a solution contact layer manufactured from a blend of polypropylene and synthetic elastomer. The back side is a clear monolayer film manufactured from a PP/synthetic elastomer blend. The drug compartment has a peelable high-barrier foil to protect the drug from moisture and/or oxygen. The high-barrier foil is peelable so that the drug can be inspected before admixture. This foil laminate consists of PET, foil, and modified ethylene vinyl acetate. Under the peelable foil material, an additional clear high-barrier material protects the drug for a limited time after the peelable foil barrier is removed. This clear barrier consists of PET with a SiOx coating laminated to a PP layer so it can be sealed to the clear monolayer.
Complex equipment
The equipment side of the Duplex story is even more complex than the construction of the bag. The line consists of four interconnected isolator modules, the first of which is called the Powder Loading Isolator. The powder arrives in 5- to 10-kg (11.023- to 22.05-lb) bulk portions packed in several different formats. These bulk portions are placed by hand in the Powder Loading Isolator via a Rapid Transfer Port (RTP). Once in the Powder Loading Isolator, the bulk packs are decontaminated with vaporized hydrogen peroxide. The number of bulk packages decontaminated at a time is considered proprietary. But the decontamination cycle can take up to 10 hours. When it’s complete, the exterior of the bulk packs and the interior of the isolator have both been decontaminated. The operator then reaches inside the isolator via glove ports, cuts the bulk packs open, and feeds the powder contents into a funnel that leads out of the Powder Loading Isolator and into the Powder Filling Isolator.
The Powder Filling Isolator receives Duplex containers from the Carrier Entry Isolator. The sterilized containers are inside carrier trays that have undergone electron-beam sterilization. An operator feeds carrier trays into the Carrier Entry Isolator by way of a tunnel that decontaminates the outside of the carrier tray by exposure to ultraviolet light. Once the tray is inside the Carrier Entry Isolator, the operator uses glove ports to peel the tray open and put a stack of Duplex containers onto a transfer belt. That belt takes the containers to the Powder Filling Isolator.
In the Powder Filling Isolator, Duplex containers are magazine-fed and everything is done automatically. Without being too precise about how the containers are manipulated, B. Braun describes powder filling this way.
First, the powder compartment of the bag is opened by vacuum cups. Powder is dispensed by a multi-cavity rotating dosing disc that volumetrically delivers precise amounts of powder from the overhead hopper. Heat-sealing follows. An integrated checkweighing system permits statistical sampling of the powder weights while the filling occurs.
Filled with powder, the Duplex container now advances automatically to the Liquid Filling Isolator. Vacuum cups pull the liquid compartment open. Then a volumetric pump dispenses the diluent into the liquid compartment and the compartment is heat-sealed. The bag drops out of the isolator and onto the conveyor that is part of a secondary packaging system supplied primarily by Concep Machine Co. (Northbrook, IL). Essentially a conveying/accumulating/collating/tray-loading system, it carries each Duplex container past a Domino Amjet (Gurnee, IL) A-300 ink-jet coder for lot and date coding.
Collating
The containers are then collated into groups of 20 and placed on a transfer system from Bosch Rexroth (Hoffman Estates, IL). This system consists of 12”-sq workpiece pallets that continuously circulate on a conveyor loop and deliver Duplex containers on demand to a station where they’re mechanically removed and sent through a wash/rinse/dry station.
Next is a Label-Aire (Fullerton, CA) labeler that applies a preprinted pressure-sensitive label to each Duplex container. On the label is a bar code that is scanned shortly after application to ensure that the correct label is in place. Then a machine vision sensor from DVT (Norcross, GA) verifies that the ink-jet code on the container is correct and that the label isn’t skewed.
The bar code on the label is also a useful tool at the customer end. “Hospitals are going to bar codes in a big way,” says director of marketing Sandberg. “They started using them for inventory management, then they went to billing patients with an assist from a bar code, then they moved to charting the patient’s record by bar code, and now they’re using them to help prevent medication errors.”
Shortly after the DVT inspection system, an indexing conveyor takes the Duplex containers through a checkweigher from Hi-Speed Checkweigher (Ithaca, NY). Along the way, every 12th bag is diverted to a nondestructive leak tester developed in-house by B. Braun.
The next station in the Concep system is a folding operation, where each bag is folded in half. Then the folded bag is advanced to a station where it is picked up by a mechanical gripper and placed in a plastic tray. After a dozen Duplex containers are in the tray, a plastic lid is dropped automatically and then the tray moves through a taper that secures the lid to the tray. Finally, two trays are packed by hand in a corrugated shipper.
July 2001 marked the first delivery of Duplex containers to customers. But equipment design began as far back as late 1994, which was when B. Braun selected IMA for the difficult task of developing this complex filling system.
The time and energy spent developing the Duplex container system have apparently been well spent. According to Sandberg, the package has been extremely well received in the marketplace, and the line now runs in full-production mode.
“It’s not just a convenience benefit we bring with this package,” says Sandberg. “It’s a safety benefit, too. By combining a fixed dose of diluent and drug in the same package, now there’s no way to make a mixing error and no way for contamination to enter the picture.”