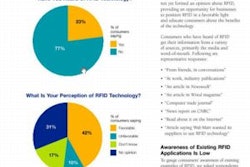
Further, HDMA is calling for pharmaceutical packagers and manufacturers to use the technology on selling units by 2007.
The EPC identifies the product’s code, lot number, and a serial number. Because it’s embedded into a microchip that is part of the tag, it allows the product information to be “read” at each distribution point all the way to the provider or retail outlet. Enforcement of the RFID marking is considered voluntary.
Several HDMA members have vocally supported the goal, including drug wholesalers AmerisourceBergen, McKesson, and Cardinal Health. Cardinal calls it the “best and most effective way for our industry to provide additional layers of security and functionality,” says Mark Parrish, Cardinal’s group president of pharmaceuticals.
To support the initiative, HDMA has issued a white paper, “Protecting Safety and Improving Efficiencies in the Supply Chain—Building a Case for RFID Technology,” drafted by the group’s Collaborative Commerce Committee. The white paper is available on the organization’s Web site, www.healthcaredistribution.org.