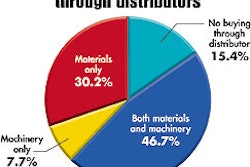
So says the first supplement to The United States Pharmacopeia, 25th Revision, and the National Formulary, 20th Edition, 25th revision. The information does not carry legal force nor does it replace requirements from any regulatory agency.
For drugs that require moisture protection, the supplement recommends that repackagers like hospitals and clinics use a blister film that provides high or extreme moisture protection. The chapter rates a number of blister materials, suggesting that polyvinyl chloride provides only a nominal-to-zero moisture barrier.
However, when laminated to a fluoropolymer or coated with polyvinylidene chloride, PVC can achieve an extreme level of protection. The level of protection, a chapter points out, should be judged on the material’s performance as a formed blister, not as a flat sheet. The chapter also suggests how to calculate “beyond-use” dates and an inspection program for the repackaging operation. —AO