After enjoying years of prosperity, Ireland has been hit by the challenging economic times that have touched many countries around the globe. In the opinion of the managing director of one medical device sub-contract manufacturing/packaging company, “things are likely to get worse before they get better” in Ireland. The good news: Through government-based groups such as Enterprise Ireland (www.enterprise-ireland.com), there is a commitment to financial investments in the life sciences sector, student education, and workforce development.
In late August, Healthcare Packaging visited four packaging-centric firms in Ireland: The Perigord Group (www.perigordgroup.com), Dublin; Sealed Air Nelipak (www.nelipak.com), Galway; Contech Ireland (www.contechireland.com), Galway; and SteriPack Medical (www.steripack.ie), Clara.
Managing global packaging artwork
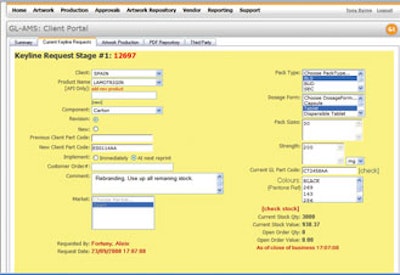
Some of the world’s most recognizable generic pharmaceutical product manufacturers are now employing the GL-AMS (Global Artwork Management System) from The Perigord Group (see photo, above right).
“The pharmaceutical sector represents between 40 and 50 percent of our business today, and that figure continues to grow,” says Alan Leamy, Perigord’s managing director. GL-AMS was introduced in the U.S. during Interphex, held earlier this year in Philadelphia. GL-AMS creates a central online storage area that provides a single point of proof for artwork used for packaging materials such as cartons, labels, inserts, and leaflets, allowing users to develop consistent branding across multiple stock-keeping units and at multiple global locations. This can help speed product to market and assist with verification tasks.
“There’s been a softening in the economy recently, but growth looks promising for us, particularly in the films side of our business,” says Sean Egan, Nelipak’s business development manager. The company provides contract packaging and assembly, validation, design, prototyping, and automation, as well as the manufacture of rigid and flexible plastics, trays, cartons, and labels.
“We don’t see ourselves as much as a supplier any more, but as a resource,” Egan says. One example of this is in the growing biomedical area, in which Egan explains that Nelipak will focus on understanding the impact of regulatory issues as they relate to biomedical products. The company will then be able to employ that knowledge to assist customers, as opposed to relying on customers to advise Nelipak on such regulations.
For years, presenters at pharmaceutical and medical device packaging events have spoken of partnering with suppliers. Nelipak and some of its customers are taking partnering to a deeper level. “About 60 percent of Nelipak employees have gone to customer sites for visits to learn more about their operations,” says Egan. “Customers love this approach because they realize we care about their process—we’re not just about providing them with a product. We also invite customers to see our plant and view our site. Here, they can see training records of our employees to learn their skill competency levels.”
Packaging for heart disease,
cancer treatments
Not far from Nelipak, Contech Ireland’s facilities are located in another industrial park that’s peppered with medical device manufacturing facilities. Medtronic, for example, is said to employ thousands of people at multiple buildings within the town of Galway. Contech provides single-use high-density polyethylene and larger-diameter polypropylene carrier tubes, used by hospitals in cardiovascular procedures. “These are high-volume dispensers that help carry stents and balloons, fluid delivery systems, and accessory pouches,” explain Debra O’Loughlin, managing director, and Lorraine Eagleton, sales and marketing manager.
O’Loughlin points out that costs continue to be a key issue for Contech customers, as do regulatory matters. A growing business area for Contech, which also provides assembly and contract services, is in oncology, where its winged infusion sets are used for delivery of chemotherapy and pain relief treatments. At the company’s 35,000-sq-ft facility, Contech operators semi-automatically assemble products in three Class 100,000 clean rooms and one Class 10,000 clean room.
Contech recently purchased a tube extrusion machine from ASG Medical (www.asgmedical.com). It expects the machine to cut customer lead times to two to three weeks, down from four to six weeks. Also important to Contech’s operation are a sealer from Riverside Medical Packaging (www.riversidemedical.co.uk) and a UV table used to cure adhesives, which is supplied by Loctite (www.loctite-europe.com).
Advanced medical
device bag introduced
At SteriPack Medical, business has led to continued expansion of the company’s manufacturing facility in Clara. Sales director Barry Moore demonstrated a customer’s new medical device package, which could not yet be discussed, and the flexible structure SteriPack provides for the thermoformed blister pack.
The company is launching an advanced version of its medical Steribag that includes DuPont Tyvek (www.tyvek.com) sealed to linear low-density polyethylene film. SteriPack describes the Steribag as similar to a conventional header bag that combines both Tyvek and film in the header strip, offering a peelable structure with breathability and economic savings. Designed for large or bulky items, as well as surgical kits and drapes, the Steribag is ethylene oxide- and gamma-sterilizable. Steribags are produced in a Class 100,000 clean room.