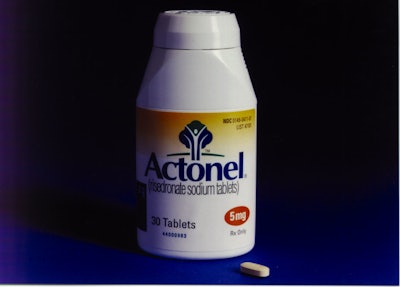
In what may be a first since birth-control pills were supplied to pharmacies prefilled into compacts, Procter & Gamble Pharmaceuticals, Mason, OH, is offering its Actonel® prescription medication in 30-count filled and sealed bottles.
“We felt that the containers normally used for prescription drugs were far more child-resistant than they were senior-friendly,” says Kurt Trombley, a senior packaging engineer at P&G. In ’94, P&G introduced a version of Scope mouthwash and later its Aleve analgesic tablet in containers that are topped with P&G’s own Safety Squease™ closure that was made by West Pharmaceutical Services (Lionville, PA). The polypropylene closure is a squeeze and twist cap that reseals with an audible click (see Packaging World, Feb. ’95, p. 72).
“We thought there was great potential to take a technology that we’d already developed and move it into the pharmaceuticals area,” says Trombley. Much like before its introduction in ’94, the closure was the subject of extensive focus group testing. “People preferred it,” he says. Testing was also conducted with both physicians and with pharmacists.