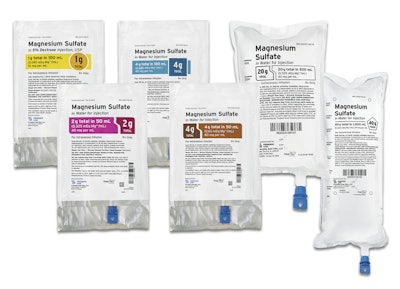
Fresenius Kabi introduced six presentations of Magnesium Sulfate in convenient, ready-to-administer Freeflex® bags.
As an integrated company, Fresenius markets both the bag and the product.
Magnesium Sulfate is indicated for the prevention and control of seizures in preeclampsia and eclampsia. Preeclampsia and eclampsia are pregnancy-related conditions that involve the development or worsening of high blood pressure during the second half of pregnancy.
Freeflex is a proprietary product designed, developed and manufactured by Fresenius Kabi. It is made of multilayer polyolefin film and is non-DEHP, non-PVC and latex-free. It is designed for easy handling with enhanced safety and environmental performance in mind. It is used for infusion solutions with its patented port technology, clear labeling and innovative product concept.
Fresenius Kabi Magnesium Sulfate in Sterile Water for Injection is now available in the following five Freeflex presentations:
• Magnesium Sulfate 4% Freeflex 50 mL; 2 gm/50 mL,
• Magnesium Sulfate 4% Freeflex 100 mL; 4 gm/100 mL,
• Magnesium Sulfate 4% Freeflex 1000 mL; 40 g/1000 mL,
• Magnesium Sulfate 4% Freeflex 500 mL; 20 g/500 mL,
• Magnesium Sulfate 8% Freeflex 50 mL; 4 gm/50 mL.
Additionally, Fresenius Kabi Magnesium Sulfate in 5% Dextrose, USP is now available in a 1 gm/100 mL presentation.
All six presentations are new offerings from Fresenius Kabi and are available immediately for U.S. customers. The products feature differentiated labeling that can help providers distinguish between different strengths. Fresenius Kabi also offers Magnesium Sulfate in four vial presentations.
Fresenius Kabi is a global health care company that specializes in medicines and technologies for infusion, transfusion and clinical nutrition.
These new offerings reflect the growth in demand for the company’s ready-to-administer products.
Magnesium Sulfate is the fifth premix presentation Fresenius Kabi has introduced in the U.S. since 2013 using the company’s Freeflex drug delivery system.
The Freeflex delivery system has been marketed globally since 2005 and in the U.S. since 2013.






















