Every person deserves access to medicine when they are sick, regardless of race, ethnicity, political stance, religious affiliation or economic status. And when emergencies erupt—like the recent California floods, or the devastation in Haiti from Hurricane Matthew, or when war or famine turn into human tragedy as has been happening in Syria and South Sudan—there must be a way to deliver the medical supplies that victims need to survive. That is where Direct Relief comes in.
Direct Relief is a nonprofit humanitarian aid organization that is active across all 50 of the United States and more than 80 countries. The organization provides medical supplies to health centers and clinics throughout the world at no charge. In the U.S., it operates the largest charitable medicine program, working with 100 medical manufacturers that donate drugs and supplies.
In fact, it is the first and only U.S. nonprofit to be certified by the National Association of Boards of Pharmacy (NABP) as a Verified-Accredited Wholesale Distributor (VAWD). The VAWD program provides assurance that the wholesale distribution facility operates legitimately and is employing security and best practices for safely distributing prescription drugs from manufacturers to pharmacies and other institutions. As a result, Direct Relief must abide by U.S. Food and Drug Administration (FDA) regulations.
“We are a wholesale distributor, which means we are subject to the Drug Supply Chain Security Act,” says Dawn Long, director of information and technology at Direct Relief, based in Goleta, Calif. “That means we are required to start complying with the elements of the law that are about the traceability of products.”
 |
| In the days following Hurricane Matthew’s landfall in Haiti, medical aid from Direct Relief arrives by ship and is offloaded near Les Anglais, on the country’s southern peninsula. |
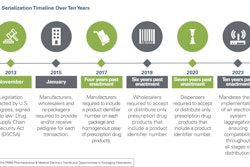
Whether it’s a charitable organization like Direct Relief or a for-profit pharmaceutical manufacturer, contract manufacturing organization (CMO), distributor or repackager, there are upcoming serialization deadlines to adhere to. Specifically, by November of this year, the Drug Supply Chain Security Act (DSCSA) requires that manufacturers mark item-level packages—or units of sale—with a unique product identifier, lot number and expiration date (lot-level tracing was required to be in place in 2015). By November 2018, repackagers must comply with the same serialization mandates, with distributors facing their first serialization deadline in November 2019 and dispensers in 2020. Ultimately, by 2023, there must be full product tracking down to each unit and an interoperable electronic system for traceability across the supply chain.
The goal of the DSCSA is to facilitate the exchange of information at the individual package level about where a drug has been in the supply chain. This enables verification of the legitimacy of the drug product identifier, enhances detection and notification of counterfeit drugs and enables more efficient recalls.
All in the industry agree that getting bogus products out of the supply chain and expediting recalls is important. But the question from supply chain partners now is: What do we do to comply?
“The first thing our customers are seeking is clarity,” says Rob Magee, portfolio director at Vantage Consulting Group, which specializes in serialization integration for pharmaceutical contract manufacturing and packaging organizations. “The end result, technically, will be a fully serialized supply chain.”
The FDA is supposed to provide ongoing guidance, according to Magee. But the FDA has issued the mandate without specific marching orders. They tell you what you need to do, “but they never tell you how to do it,” Long says. “The industry has to work together to come up with a solution that will work for everyone.”
Indeed, it is up to industry to figure it out. And therein lies the biggest obstacle of all, because although there are a variety of approaches and new groups emerging that will help integrate current manufacturing, packaging and supply chain standards, there is not a universal method for exchanging serialization data in the supply chain—only suggested options.
On top of that, every country takes a different approach to serialization, which means how you identify products and the type of information required for each transaction could vary depending on where in the world products are shipped.
For example, in China, the government generates serial numbers and everything must be reported back to them. The European Union (EU) model allows manufacturers to create serialization data, but it too must be recorded in the centralized EU hub. The U.S. is focused on a chain of custody, where manufacturers and supply chain partners generate and own the information, leaning more toward liability than visibility in the track-and-trace structure. In the U.S.-based chain-of-custody approach, a product can ship to 50 different locations and it is not tracked, except when it reaches the possession of another owner.
“It is a handshake of trust around who owns [the product] and who is responsible for it,” says Dave Harty, vice president of professional services in the Americas for Adents, a maker of unit identification and traceability software. “But the minute you don’t own it, you need a transaction history and trail.”
In the case of Direct Relief, the organization gets advanced shipping notifications from a manufacturer, or the organization will capture lot numbers at receiving and store that in a system from Axway to trace that data to wherever it will be sent to next. The system also creates a cloud portal for partners to see the chain of custody, Long says.
This model really does resemble a handshake, Harty says. The problem here for the industry as a whole is that there is no uniform way to exchange that chain-of-custody data. “It’s not that there are no standards; in fact, there are multiple standards, and that’s the problem,” he says.
For example, a model from GS1 provides a lot of mechanisms to identify product data, all of which are acceptable to the FDA. “They haven’t ruled anything out and there are no limits or restrictions on anybody, which creates a conundrum,” Harty says.
The GS1 way
GS1 US is a not-for-profit standards organization—with more than 300,000 members across 25 industries—that provides a common language to help create seamless work processes, thereby enabling businesses to identify, capture and share information the same way around the world. The GS1 standards focus on identification numbers that uniquely distinguish products, logistics units, locations, assets, documents and relationships across the supply chain from the manufacturer to the consumer.
The GS1 Global Trade Item Number (GTIN), for example, identifies trade items as they move through the global supply chain. The GTIN is placed in a barcode or Electronic Product Code (EPC) to ensure the same information contained in the corresponding electronic documents are being processed between trading partners. A Global Location Number (GLN), encoded in either a barcode or EPC/RFID tag, identifies where a product is shipped and can identify physical locations like a warehouse or a port, or can identify a company’s function as a buyer, seller or carrier. Electronic data interchange (EDI) provides common messaging and master data alignment around order, delivery and financial settlement. Similarly, the Electronic Product Code Information Services (EPCIS) creates and shares event data about the physical movement and status of products as they travel through the supply chain. And the serial shipping container code (SSCC) can be used to identify a logistic unit, like a case, pallet or parcel.
“Standards identify, capture and share data,” says Peter Sturtevant, senior director of industry engagement for GS1 US. This provides pharmaceutical manufacturers with the tools to create global trade item numbers for each product right down to the lowest saleable unit, as will be required by law. “But the brand owner, the manufacturer, has a significant responsibility to make sure they are trading information properly and accurately.”
To help in that effort, GS1 US published a new release of the guideline, Applying GS1 Standards for DSCSA and Traceability (R1.2), which builds upon version 1.1 and includes new features in the EPCIS and Core Business Vocabulary (CBV) standards. The guideline is being published in the GS1 US DSCSA Implementation Suite, which includes three reference documents containing additional guidance on business process applications.
In addition to this guidance, however, manufacturers and CMOs will need a way to manage the process. That’s where technology providers like Adents and TraceLink can help.
Adents’ Pharma Suite includes centralized configuration and process management software. It can handle setup parameters for multiple lines, managing item, bundle, case, package and aggregation. It also interfaces with enterprise resource planning (ERP) and manufacturing execution systems (MES) to collect work orders, as well as work with GS1’s EPCIS. A supervisor component centralizes all data in real time and can generate production reports or exchange serialization data with supply chain partners. In addition, Adents recently partnered with Microsoft to create a cloud platform, called Adents Prodigi, that can centrally manage regulatory requirements and give pharma labs and CMOs the ability to track a unique product identifier on a global level.
TraceLink has technology to generate serial numbers and send them to packaging lines. It can also exchange serialization data between business partners, supporting GS1 standard formats and generating custom formats for places like Brazil or importing custom formats and lists from China, for example. The TraceLink Life Sciences Cloud is a globally networked track-and-trace ecosystem that uses Amazon Web Services and handles regulatory requirements and serialization data for anywhere in the world by converting data to formats that trading partners use. Authorized users can update, monitor and report transactions in real time so that supply partners work together.
“We focus first on building a network that everyone can connect into and do so using industry standards,” says Brian Daleiden, vice president of industry marketing at TraceLink. “Then we focus on building the apps needed to meet requirements under the law.”
Open-SCS on the line
The vendor community understands the importance of aligning with standard-setting groups like GS1, which is why serialization companies like Adents and TraceLink are also members of a new working group, the Open Serialization Communication Standard (Open-SCS), which is trying to tackle data exchange issues from yet another angle—interfaces between devices and controllers on the packaging lines. While GS1 standards focus on supply chain interfaces, Open-SCS is trying to solve serialization interoperability challenges inside the organization, because of all the heterogeneous systems from multiple vendors that don’t necessarily integrate easily with each other.
Though there are existing plant-floor integration standards, like ISA-95/88, Business to Manufacturing Markup Language (B2MML), OPC UA and Packaging Machine Language (PackML), there needs to be new specifications and guidelines to get these standards working together for serialization purposes.
“Open-SCS is a way to standardize communications up and down the different layers of packaging execution systems within the four walls of an organization,” says Open-SCS member Dirk Rodgers, a regulatory strategist with Systech International and the founder of RxTrace. “Too many people think we are trying to work on something that will compete with GS1 standards, but that is not true.”
In fact, Open-SCS, in partnership with the OPC Foundation, is collaborating with GS1 EPCIS to create a seamless information exchange from packaging line operations to the supply chain. In addition, the group is working on a Packaging Serialization Global Name Space Registry to develop a set of standard information-exchange specifications.
“On our docket of things to do this year is the Global Name Space Registry, which will be an open-source registry,” says Charlie Gifford, technical director of Open-SCS. The main issue it will solve is the dilemma of having to meet the reporting requirements of different countries, where there are not just data translation issues, but language issues as well. For example, an English translation of a Chinese regulation might not always be accurate when it comes to reporting requirements.
“We will try to build a database to register what the reporting elements are that have to come out of the plant for a given country and a given regulation,” Gifford says. “And once we build that, we will make sure our interfaces support data collection at the line and the site systems and the enterprise.”
That means a large pharmaceutical company with five lines in five countries by five different vendors doesn’t have to figure out the data mapping, but can just apply the Name Space Registry and the certified interface.
Of course, even with all the work that Open-SCS and GS1 are doing, there are still weak links in the supply chain—including establishing a trusted and secure way to do business with independent trading partners and other contract organizations that might have been hired for a specific project. This is where some industry observers are pointing to blockchain technology, an unalterable peer-to-peer recordkeeping system that enables communities to securely record and share information. Blockchain is best known as the core component of the digital currency Bitcoin. Each validated transaction creates a block, which attaches to the chain of blocks before it, thereby creating an easy-to-follow trail.
The blockchain discussion is still very much in its infancy and, perhaps, it’s a technology best kept on the back burner for now, as the industry faces a more pressing issue of the looming November deadline.
“Today, DSCSA is lot-based and it is estimated that only around 10 percent of prescription drugs are serialized,” GS1’s Sturtevant says. “By November 2017, it should be at 100 percent.”
Stepping back from the technology struggles the DSCSA regulations represent, perhaps the industry does indeed need a deadline to deliver what the world wants: safe medicine.
“At the end of the day, this will make medicine safer for people,” says Direct Relief’s Long. “Just because we offer charitable medicine doesn’t mean we will provide lesser quality than a paying customer. But the reality is that we are not doing any serialization now and it really is a big deal. The industry is coming together and working on what those standards are, and once it is fleshed out, we will [all] react better.”