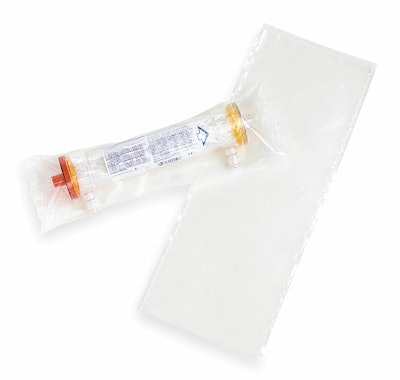
Dialyzer package
Highest Achievement—Packaging Excellence
Gold—Environmental Achievement
Gold—Technical Innovation
Baxter International, Deerfield, IL, benefits from rollstock material that it uses for horizontal form/fill/seal of a dialyzer, a medical device used in hospitals or in dialysis clinics for patients with end-stage renal disease.
Rollprint supplies a 3-mil structure that it describes as a special polyethylene blend that offers durability and puncture resistance. Rollprint machine-punches holes into the film to make it breathable, which is necessary as the pouches must withstand ethylene-oxide sterilization.
That created a challenge in making the structure. “If we had a film that was too puncture-resistant, we couldn’t uniformly needle-punch the holes in a consistent pattern,” explains Rollprint’s Karen Berger. “We needed to have enough porosity in the film to enable our customer to go through the same sterilization process as before, and to go through EtO sterilization in a consistent manner. If the holes in the film are too large, then they could let dust and small particulate matter in, which would not be desirable.”
Before using these tubes, Rollprint supplied Baxter with premade pouches. “The premade pouches were three-side-sealed and we put product in them before heat-sealing them,” recalls Cathy St. John, a packaging engineer in Baxter’s Renal Division in McGaw Park, IL. “But we’re always working to try to keep costs down so that our pricing to customers can remain the same. The premade pouches were expensive.”
She notes, “Rollprint has been a wonderful supplier to Baxter over the years and they’re just phenomenal people to work with. By design, we knew the packaging material had to breathe so that during EtO sterilization the gas could get in and out of the product. Once we knew what we wanted, we went to Rollprint and asked them to create it. We’ve been using it since mid last year.”
St. John says the devices are packaged at a Baxter plant in Arkansas and she says the ability to automate the packaging process has created savings, despite the purchase of a new hf/f/s machine. “With premade pouches we had to have handlers on the line to open the pouch, insert the product, then put it through a sealer,” she says. “Now we convey product down a line and it’s automatically filled. We only need one individual to place them in a shipping case. The labor savings make it [the cost] come out equal.”
The unprinted film pack measures approximately 16” L x 6” W, St. John estimates. “It’s not a sterile package,” she notes. “It’s sterile fluid pass. The device has protectors on the fluid pass to ensure that if they remain in place, once it goes through terminal sterilization you guarantee the fluid pass is sterile, but the outside of the pack is not.”
Since the device has been marketed globally in the new pack, customers have noticed. “Everyone’s been pleased with it, and it’s 100-percent recyclable,” St. John says. It’s helped Baxter reach its goal of always trying to reduce costs so that customers don’t incur a cost increase, she adds. —Jim Butschli
Sterilization pouch for surgery
Gold—Technical Innovation
In the Autopak® Sterilization Pouch, two of the three layers used to make the pouch are supplied by Alcan Packaging. A 4-mil outer clear top ply is an adhesive lamination of DuPont Canada’s nylon with a polypropylene-based film produced by Alcan at its Asheville, NC, plant. This portion of the structure provides transparency and abuse-resistance for large medical devices and trays containing smaller devices.
The middle ply, made at Alcan’s Marshall, NC, facility, is a PP-based multilayer coextrusion perforated for permeability. It contains an autoclavable peelable seal feature, which activates after steam sterilization.
The outer bottom ply is a spunbond-meltblown-spunbond nonwoven sheet from BBA Fiberweb. This part of the structure allows permeation of sterilization gases such as steam while it also provides a barrier to bacterial penetration. All total, the structure measures between 18 and 22 mils thick, “depending on how hard you compress the spunbonded layer,” asserts David Rudd, senior specialist engineer at Cardinal Health Care.
Cardinal Health converts materials into a three-side-sealed pouch that it sells to hospitals and the dental market for use in surgical applications around the globe. Only lot number identification information is printed on the pouch, Rudd says. Cardinal heat-seals together the Alcan-laminated three-layer structure at a facility in North Carolina.
In late 2003, the Autopak Sterilization Pouch replaced what Rudd refers to as a sterilization wrap. Cardinal literature touts the pouch as “an innovative new product that provides visual access to package contents, time and motion economies in preparation and presentation, and the means to reduce the total cost of instrument processing. The end user can quickly confirm whether the instruments are in acceptable condition for use. When packs are delivered to the end user, the contents are presented easily and aseptically. Finally, material waste is meaningfully reduced and disposal costs are minimized.”
Rudd adds, “Autopak is not meant to completely replace sterilization wrap, but it is ideal for specific applications. Autopak has been very well received.”
—Jim Butschli
To see packages earning FPA Silver Awards, see: packworld.com/go/w138"
See sidebar to this article: Seen Previously in Packaging World
See sidebar to this article: Low-profile, easy-open microwave popcorn bag
See sidebar to this article: Non-foil 18-oz retort pouch provides microwaveability
See sidebar to this article: Flexo-printed all-plastic pet food bags
See sidebar to this article: Shaped ‘tree’ pouch grows sales in Costa Rica
See sidebar to this article: Convenience pack is no turkey