The small electronic tags on the packaging will allow tracking of drugs as they move through the supply chain.
The agency published a compliance policy guide for implementing RFID feasibility studies and pilot programs with an eye toward having RFID in the drug supply chain by 2007. FDA has established an internal RFID workgroup to monitor RFID adoption in the drug supply chain, identify regulatory issues associated with RFID use, and develop processes for handling the issues. FDA involvement is critical because a drug company cannot change a product label without FDA approval.
Pfizer announced it would put RFID tags on all bottles of Viagra as expeditiously as possible in 2005. Purdue Pharma will tag bottles of OxyContin and, if enough tags are available, Palladone. GlaxoSmithKline said it would begin using the tags within the next 12 to 18 months on one of its drugs believed susceptible to counterfeiting. Initially, the tags will be placed only on the large bottles shipped to pharmacists to fill individual prescriptions.
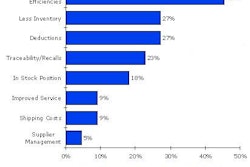
Each label costs 20¢ to 50¢, with readers and scanners costing thousands of dollars. Prices are expected to fall once RFID tags become more popular and in wider use. Labels will remain voluntary until 2007; after that, FDA could mandate their use and the type of tag.