Rockville, MD-based biotechnology company Sanaria’s website says its “primary mission is to develop and commercialize whole-parasite sporozoite vaccines that confer high-level, long-lasting protection against Plasmodium falciparum, the malaria parasite responsible for more than 95% of malaria associated severe illness and death worldwide and the malaria parasite for which there is the most significant drug resistance.”
Distribution of vaccines into the developing world continues to be a significant challenge to disease prevention efforts. Sanaria and Cryoport are actively working toward development of a robust distribution network through use of a liquid nitrogen vapor phase (LNVP) cold chain, which both believe is critical to the success of vaccine/ biologics distribution throughout the African subcontinent, and provides significant advantages over current biologics delivery systems.
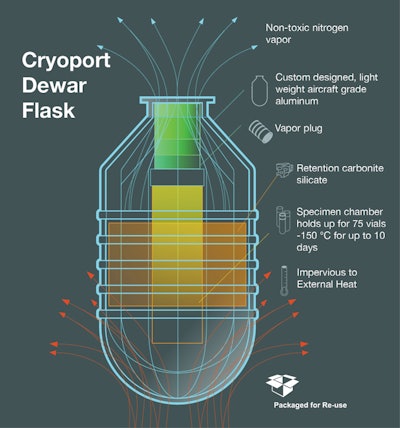
(Shown in the illustration is a schematic of Cryoport’s liquid nitrogen dry vapor shipper.)
Dr. Stephen L. Hoffman, Sanaria’s Chief Executive and Scientific Officer, says, “Our mission is to develop a vaccine that will combat a malaria parasite that is the most drug-resistant and causes more than 98% of malaria related deaths worldwide. With Cryoport as our logistics partner, we can trust that their validated cold chain logistics solutions will provide a reliable and effective distribution strategy to bring to market an effective preventative vaccine for this deadly disease. We also rely on Cryoport for delivery of our family of products that are intended to advance and promote high quality research for new vaccines and drugs.”