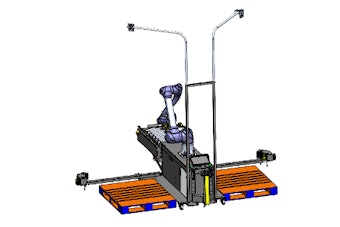
Next Generation Pharmaceutical’s (NGP) report headlined, “Are blockbuster drugs dead in the water?” illuminates a trend that could bring “disruptive” change to the pharmaceutical industry: Patent expirations and advances in the human genome and personalized medicine could lead to a more fragmented market, says NGP.
Should pharmaceutical firms begin to switch their focus from producing blockbuster drugs to emphasizing treatment for targeted groups of individuals, it’s a good bet that packaging functions will also evolve. In many cases, it’s likely that packaging lines will likely need to be more flexible and offer quicker changeover to accommodate smaller runs. That could pose quite the conundrum for pharmaceutical firms trying to boost overall equipment effectiveness and reduce costs in their packaging departments.
Another change that could develop out of this scenario is an improved relationship between pharmaceutical companies and the U.S. Food and Drug Administration. For example, the NGP article quotes Cleveland Clinic’s Dr. Steven Nissen saying, “Harder-to-develop, targeted therapies will be part of the next wave in patient care…U.S. government should create incentives to develop drugs for such treatments…if we encourage that kind of regulation, then regulation becomes [the pharmaceutical industry’s] friend rather than the enemy.”